Untitled
Particle Size Analysis by Laser Diffraction Introduction 3. The scattering pattern at the detectors is the The underlying assumption in the design of laser sum of the individual scattering patterns diffraction instruments is that the scattered light pattern formed at the detector is a summation of the scattering pattern produced by each particle Experimental that is being sam

 SEMINARS IN RESPIRATORY AND CRITICAL CARE MEDICINE/VOLUME 29, NUMBER 5
Table 1 Nomenclature for Mycobacterium aviumComplex (MAC) Organisms
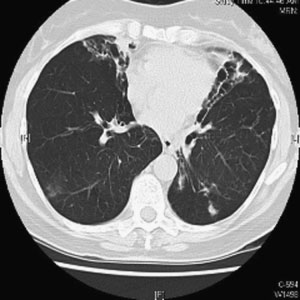
Figure 1 Chest computed tomographic scan. Right upper
lobe cavitary opacities in 63-year-old man with Mycobacter-
ium avium complex infection and underlying emphysema.
SEMINARS IN RESPIRATORY AND CRITICAL CARE MEDICINE/VOLUME 29, NUMBER 5
Table 1 Nomenclature for Mycobacterium aviumComplex (MAC) Organisms
Figure 1 Chest computed tomographic scan. Right upper
lobe cavitary opacities in 63-year-old man with Mycobacter-
ium avium complex infection and underlying emphysema. DIAGNOSIS AND TREATMENT OF INFECTIONS DUE TO MYCOBACTERIUM AVIUM COMPLEX/KASPERBAUER, DALEY
with tuberculosis, isolation of a single positive sputumculture does not necessarily represent disease so diag-nostic criteria have been developed to aid the clinician indeciding whether treatment is indicated. Unfortunately,well-designed and appropriately powered studies for thetreatment of immunocompetent hosts are still lacking.
DIAGNOSIS AND TREATMENT OF INFECTIONS DUE TO MYCOBACTERIUM AVIUM COMPLEX/KASPERBAUER, DALEY
with tuberculosis, isolation of a single positive sputumculture does not necessarily represent disease so diag-nostic criteria have been developed to aid the clinician indeciding whether treatment is indicated. Unfortunately,well-designed and appropriately powered studies for thetreatment of immunocompetent hosts are still lacking.